Firstly, use this link as I personally find it far superior to using the wiki (I might be biased):
You can:
- Search for chemicals much faster than the wiki
- Click through to other chemicals in a faster way
- Use the back/forward buttons in your browser to quickly navigate to pages you were just on (super useful for getting back to the chemical you started on before diving)
- Click the “Tree View” button to show a recipe and all its sub recipes/requirements on a single page
- See friendly upfront warnings for certain reactions
tl;dr
- Use the app and ensure your Starting pH (S:) and Ouput Chemical pH (O:) are okay. If they’re red, take action BEFORE your reaction, rather than trying to spam buffers whilst it’s reacting
- Do reactions in smaller steps, adjusting pH as you go. Don’t just dump 20u of every required chem in a container and then struggle to control it. Do it in stages or cool the reaction down to slow it down.
- Be sure to use the Chromatography machine to check Purity often when you’re learning. Do NOT rely on this to make things pure (it’s slow and a crutch and cannot get to 100%)
- Your Oil, and other common chems, are probably 66% impure and making any chemicals you use that in impossible to make pure and very easily turning your reactions in to sludge.
- Roundstart chemicals/medicines can have impurities in them!
Who is this Guide for?
Anyone who already understands the basics of chemistry, dispensing chems, reactions, etc. but is struggling with understanding pH, purity and/or always getting inverse/failed chems.
For example, are you trying to make something and always getting Vicious Sludge? Or is the chromatography machine telling you you’ve got a ton of impurities?
Intro
This guide will only focus on pH and how to control it - as it is the main area I see people struggle with. I may write more general chemistry guides at a later date.
What is pH
We’ll ignore real life as it just confuses things and focus entirely on how it works on Fulp/Tg.
It is used to represent how acidic/basic a chemical is. It’s a linear scale from 0-14, with 0 being Acidic and 14 being Basic.
It’s main purpose is to affect the outcome of reactions (turning one set of chemicals in to another chemical) by impacting the purity of the reaction.
Measuring pH
Every chemical has a natural pH, this is the pH of the chemical if it was created out of thin air, such as spawning in bottles, products from vendors, dispensed from a machine, etc. Chemicals created via reactions will very likely NOT have their natural pH, more on that later.
Shift Clicking a container with the Chemistry Goggles on will tell you the pH of the container.
A containers’s pH is technically a separate measurement but it is also the more important measurement as that is what is used for reactions. If a container only has ONE chemical in it, the pH reported is the pH of that specific chemical. If a container has multiple chemicals then read on.
A beaker’s pH can be calculated as: The sum of each chemical’s capacity multiplied by that chemical’s pH, divided by the total volume of the beaker. E.g.
10u of Carbon (pH 5) = 10 x 5 = 50
20u of Oxygen (pH 9.2) = 20 x 9.2 = 184
5u of Hydrogen (pH 0.1) = 5 x 0.1 = 0.5
pH of container = (50 + 184 + 0.5) / 35 = 6.7
This means you can predict the pH of any mix of chemicals which is a very important skill to learn.
Purity
When a reaction takes place and the output chemical is created in the beaker. The current pH of the container will be used to calculate the purity of the reaction.
Every reaction has a minimum and maximum optimal pH, any time the output chemical is created whilst the container’s pH is outisde of this range will make the reaction impure. The further away from the optimal range, the more impure it becomes.
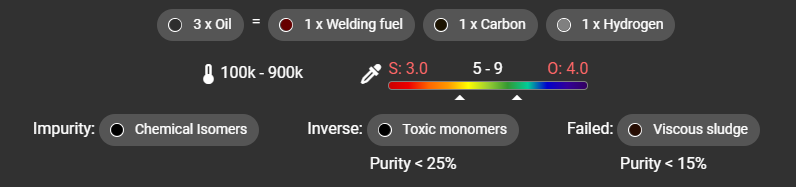
The pH range can be found on the wiki or my app:

This is saying the containers pH must be between 5-9 whilst the reaction takes place, ignore the S: and O: for now, that is covered later.
Note: A reaction is not instant and every reaction has a different effect on the pH, this means it’s important to ensure your reaction starts pure but also stays pure. Every tick the solution is in a reactable state, it will react.
Every reaction also has a value that controls which way the pH shifts as well as the pH of the output chemical affecting the container’s pH. Some reaction’s barely move pH whilst others will sky rocket to impure!
It’s also very important to note that impurity carry forwards, that is to say, if you make Oil that is only 33% pure, that Oil will ruin the purity, regardless of pH, of any reactions that Oil is used in. I find this is actually the main issue chemists have. All of their reagents being impure, stacking in to more and more impure reagents, causing the final, more difficult chems, to come out as sludge/very impure.
Impurity, Inverse and Failed Chemicals
Chemicals can have the following:
- Impurity: A chemical that will exist as part of your main chem (note that when inspecting the beaker, impurity looks like the actual output. Only the chromatography machine can tell you the exact breakdown!)
- Inverse: A chemical that replace your main chem at the end of the reaction, if the purity is below this threshold
- Failed: A chemical that will replace your main chem at the end of the reaction, if the purity is below this threshold
The purity values, the chemicals and if they even have these are defined per chemical.
Note: even round start/spawned/bought chemicals have impurities in them! Ever wondered why using Aiuri (the burn heal) makes your vision blurry for a while? That’s because it’s impure and has some Aivime in. It’s the Aivime that does the blurriness! If you are able to make 100% pure Aiuri, it does not cause blurriness (though Aiuri will still do eye damage).
Using my app, you can view the impurity section as part of the reaction:

These reads as, when the reaction happens, if the purity is not 100%, some of the output chem will actually be the impurity (Chemical Isomer in this example). If the purity of the overall reaction falls below 25% but above 15%, the solution will become the Inverse (Toxic Monomers), if the reaction’s overall purity falls below 15%, the solution will become the Failed (Viscous Sludge).
Controlling Reactions Tips
So now you know what pH is, what pH does and what happens when you do purity goes wrong - how do you do it right?
Before reading the blow tips, familarize yourself with the O and S values from my app:
- S: Estimated Starting pH of the reaction. This number represents an estimated pH the reaction will start at. It is very important to ensure your starting pH is in the right range otherwise you will not be able to make it 100% pure, the reaction must start pure.
- O: Estimated Output pH of the generated chemical, NOT the overall reaction. With how chemistry works, the pH of the generated chemical is very important as reactions will heavily trend towards this value! This means that if the O value is outside of the Optimal range you will need to take extra steps to ensure the reaction stays pure, otherwise your container will rapidly go impure.
Both of these values go red if they are a problem. I find these are THE main thing I use to help me with chemistry.
Adjusting pH with chemicals
This is when you take a chemical that is NOT part of the reaction but introducing it in to the beaker before the reaction starts to help control the pH. As a container’s overall pH is the average of all the chemicals in it, you can sway the pH one way by purposely using various chemicals. Hydrogen for example is great at lowering pH whilst things like Sodium are great at raising the pH.
Adjusting pH with buffers
Buffers are instantly consumed when added to a beaker but they adjust the pH (towards 0 with acidic buffer, towards 14 with basic buffer). You can use the reaction chamber or a dropper to slowly adjust the pH. This is useful both before and during a reaction.
Slow down how fast a reaction’s pH changes
This is nearly always due to the output chemical being made having a pH quite far away from the optimal/current pH. There are many ways to control this in your favour, I’ll list a few examples now:
- Make less of the desired reaction and pad the remaining volume of the beaker with a chemical thats pH is in the optimal range and at the opposite end of output chemical, this stops the output chem having such a large impact on the container pH and makes the pH easier to control in the chamber
- Do the reaction in steps: add all but one of the chemicals, balance the pH to the opposite end of the output chem’s pH, then add just 1-5u at a time of the final chem, adjusting the pH between each addition untill you are done.
- Chill the reaction to just below it’s reacting temperature, this will slow down the reaction to give you more time to respond. Note that it can be diffult to use this method on certain recipes due to freezing and/or very low temperature requirements.
These will guide you in how to prepare for your reaction and what kind of steps you could take to control it.
Ensure starting pH is inside optimal range
Easiest way to help here, is to add all of the more ‘extreme’ pH chemicals (the ones furthest outside of the reaction) but not the last chemical to start the reaction. You can then use buffers to get the pH in to the right range. Try to make sure the final chemical you add to start the reaction will not push your new pH out of the optimal range.
Note: If your reaction’s temperature is above room temperature (300ish) here, this are often the easiest to control as you can actually mix all the chemicals together before the reaction starts.
Check every chemical you make’s purity in the chromatography machine
(that’s this one - usually only one exists and it’s in the Pharmacy)

This is super important and will very quickly make you in a better chemist.
Take Oil for example. Every new Chemist knows it’s Carbon, Hydrogen, Welding Fuel. Mixing all this together will work and will give you a beaker of Oil.
What people then miss is that this Oil is 33% Pure, this is a terrible purity and ruins future reactions and Oil is a component in a lot of reactions.
Why is this? Well again in my app…
As 33% pure is greater than 25%, it did not turn in to Toxin Monomers. 66% of the Oil has actually ended up as Chemical Isomers because the starting and output pH has a huge impact on the reaction. The reaction was basically NEVER in the right pH range!
Once you know this and realise just how easy it is to start introducing impurities earlier, you can start to learn your own comfortable ways to make 100% pure version of common components. This means you can be lazier down the line with purity on the more difficult chems as some impurity is okay - but impure Oil like this is what often makes reactions so easy to fail!
Case Study - Oil
As I’ve talked about Oil a lot, let’s break this down as to why it’s such a problem. Firstly it is used in 13 recipes, 7 of those are intermediate chemicals, meaning Oil is actually used in way more than 13 recipes.
Mixing it as the recipe stands (and how most people do), gives you:
30u of Hydrogen (pH 0.1) = 30 * 0.1 = 3
30u of Welding Fuel (pH 4) = 30 * 4 = 120
30u of Carbon (pH 5+ = 30 * 5 = 150
Starting pH = (3 + 120 + 150) / 90 = 3.03
Oil’s optimal pH range when reacting is 5 - 9, a starting pH of 3.03 is bad as it instantly starts reacting impure.
Oil itself has a pH of 4. This means the reaction will heavily trend towards pH 4 as it reacts. Again, the range is 5 - 9, an output pH of 4 is no good and actually pushes/keeps the reaction impure. Even if you fixed the starting pH, Oil will still end up impure.
This means Oil is actually quite a difficult chem to make pure as BOTH things are wrong with it.
Oil reactions MUST:
- Change the pH of the container before starting the reaction
- Monitor/control the reaction whilst reacting
Method One - Adjust one step at a time
This approach can be applied to a lot of reactions. With varying speeds/amounts of chemicals/buffers required. I prefer this method over dumping it all in and using the chamber because you are 100% in control.
It is basically making a large amount of your desired chemical but only a few units at a time.
First identify all reagents BAR ONE of the recipe that are the furthest away from the optimal pH.
For Oil, this gives us Hydrogen and Welding Fuel, both outside the optimal pH range.
Put equal parts of these in a beaker (but leave room for the final chemical), so for Oil that would be 30u Hydrogen (pH 0.1) and 30u Welding Fuel (pH 4) in a beaker.
Then, using the reaction chamber OR a dropper, use Buffers to get the beaker’s pH as close to the extreme end of the optimal reaction that is opposite to the output pH of the chemical being made.
So again, Oil has a pH of 4 and the reaction has an optimal pH of 5-9. This means I would use Basic Buffer to make the container’s pH as close to but NOT above 9.
Then add 1-5u of the final reagent, in this case, Carbon and monitor the pH.
If the pH drops rapidly and is nearly going inoptimal, use buffers to raise it back to as close to where you were before. If there is still plenty of room before going inoptimal, add some more of the final reagent until the pH is close to leaving the optimal pH range.
Repeat until the beaker is fully reacted.
This may sound long/time consuming but what you will eventually realise, is the steps for Oil are the same everytime. Meaning you end up being able to do it without checking the pH at each step and just rapidly clicking droppers/beakers and mixing Oil almost as fast as you could before.
Method Two - Pad with Sodium
This is my super cheaty method to get 100% pure oil every time. Instead of doing equal parts Hydrogen, Welding Fuel and Carbon, also add in an equal part of Sodium. Be sure the add the Sodium first to avoid the Oil reacting before the sodium is added.
This gives you:
25u of Sodium (pH 11.6) = 25 * 11.6 = 290
25u of Hydrogen (pH 0.1) = 25 * 0.1 = 2.5
25u of Welding Fuel (pH 4) = 25 * 4 = 100
25u of Carbon (pH 5) = 25 * 5 = 125
Starting pH = (290 + 2.5 + 100 + 125) / 100 = 5.18
So this has fixed the starting pH - but at first glance it also seems pretty close to trending below 5 due to the Oil’s pH. However, as the Sodium itself isn’t consumed in the reaction, the 11.6 pH (roughly, it will have modified slightly…) it still has under the hood, helps to fortify/offset the Oil’s affects on the container’s pH.
You can then remove the Oil with the Chem Master and you now have 100% pure Oil!
Other recipes have similar tricks. You just need to find the right chemical and the right amount. I am not going to give my out my cheat sheet for this as I try not to use it too much. The trick is just looking for high/low pH chemicals to offset the reaction, without them actually reacting with anything else in or generated in the container.